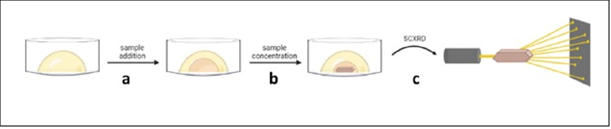
In the field of chemistry, classical methods of producing high-quality single crystals have largely evolved around time-consuming manual techniques. These methods involve slow growth solution phase crystallization based on evaporation, thermal control, or diffusion (either liquid-liquid or vapor). Such techniques suffer from inherent limitations, mostly centred around the relatively large amount of material consumed during sample preparation coupled to the significant amount of time required to produce a quality crystal. Additionally, these methods cannot give information on samples that are liquids at room temperatures.
In recent years, a number of new methods have emerged to overcome these classical limitations and facilitate crystal production for SCXRD. These can be broadly arranged into two categories, namely “host-aided” and under-oil based approaches.